The concept is logical and easy to follow--if reactions can be triggered by light, heat, pressure, or electrical potential, then why can't mechanical force also be harnessed to distort molecules in a way that promotes reaction? Actually, using molecules appropriately termed mechanophores, the Moore group has succeeded in employing the mechanical forces generated from ultrasound to promote and influence chemical reaction pathways. Although ultrasound generally has no effect on small molecules, the collapse of cavitation bubbles produced during sonication can agitate polymers in solution, generating friction, otherwise known as mechanical force. By incorporating small molecule mechanophores (either trans- or cis-1,2- dimethoxybenzocyclobutenes, BCBs) into a larger polymer, researchers were able to take advantage of this force and promote ring opening. According to the Woodward-Hoffmann rules, in a reaction promoted through light energy, both cis- and trans- BCBs undergo a disrotatory ring opening, while thermal activation produces conrotatory products. On the other hand, computational studies indicated that under mechanical influences, the cis-BCB would produce the disrotatory product, but the trans-BSB would generate the conrotatory product.
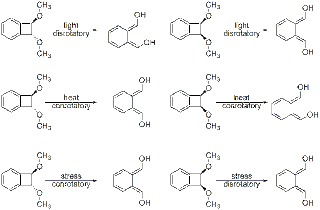
In order to test this hypothesis, the BCB-containing polymer was sonicated at 6-9 degrees C with an excess of a pyrene functionalized maleimide, which was meant to function as a a dienophile trap. Indeed, the mechanophore did behave as predicted; by distorting bond lengths and angles sonication produced the trans-BCB through a conrotatory process, while the mechanical force worked to reduce the energy barrier for a disrotatory reaction pathway for the cis-BCB. As the article title indicates, reactions can certainly be biased through the use of mechanical force.
So what will the next mechanophore be?
10 comments:
that is so cool, how those compounds react :-)
I’m impressed, I need to say. Really not often do I encounter a blog that’s each educative and entertaining, and let me inform you, you have got hit the nail on the head. Your concept is outstanding; the difficulty is one thing that not sufficient people are speaking intelligently about. I'm very blissful that I stumbled throughout this in my seek for something regarding this.
Hotels in Munich
One of the more impressive blogs Ive seen. Thanks so much for keeping the internet classy for a change. Youve got style, class, bravado. I mean it. Please keep it up because without the internet is definitely lacking in intelligence.
Naples Hotels
Thank you for your article, pretty worthwhile material.
This particular billet has been somewhat of the revelation to me.
http://www.buylovejewelry.com/
http://gamepartygogo.com/
camriver satchel is a fashion and hot bag brand, now you can buy cheap cambridge satchel at http://www.camriversatchel.org. Fluro cambridge satchel is so popular among yong people, you will like is so much.
Buy Cheap Windows 7 Key to do your office work, you will benifit a lot. windows 7 upgrade key is so vital to your office work, Office 2010 Key is also so functional, you will like them so much.
They wereDiablo III items throwing these people all around cautiously, keeping the very best element solely. From this statement I would personally state that just the tentacles mightBuy Guild Wars 2 Gold poke, however Now i'm not really a professional.
As you have just read, you will find almost certainly millions of items you didn't know about with your apple ipad. This article has presented you with a few basic tips and advice that can make this product a lot more user-friendly. Make use of the recommendations out of this and then any other post to build in your knowledge basic for that ultimate customer practical experience!IPad Suggestions You May Possibly Not Understand About [url=http://flexicareinc.org.au/V6%20Backup/includes/wp_login.asp]Air Max Australia[/url]
Most those who are sensitive to dog dander think that canines, and kitties are definitely the only animals that can induce difficulties in the house. With the exception of sea food, reptiles, and amphibians, pollen is universal among popular home critters. Ferrets, hamsters, mice, and birds are as prone to set off a hypersensitive reaction. Take this into account when you shop for your dog. [url=http://iplumb.com.au/common/client.asp]Supra Shoes[/url]
In case you are a caffeine drinker having a brief consideration span, then consider receiving a single glass producing unit. These machines pre-calculate their grounds to the best glass each and every time and also supply you with the collection of transitioning your make with every single glass you ingest. Just one-cup device also helps to ensure that each and every glass you possess is hot and freshly made. [url=http://v6.com.au/signatures/ByronLogin.htm]Abercrombie And Fitch[/url]
Diabetic issues does not have a lot of signs and when you do not acquire your medications, there is certainly typically no immediate result. However, it is quite risky as with no treatment diabetic issues can readily cause a lot more risky medical conditions such as heart disease. In case you have diabetes, make sure to stay aware about consuming your prescription drugs to prevent further complications. [url=http://abbeydigital.com.au/ixs/gallery.asp]Tiffany And Co[/url]
New studies have shown that consuming meals high in omega-3s can help with joint inflammation. Should you be not keen on seafood and fish, then you can definitely nonetheless make use of omega-3s by taking a daily omega-3 fatty acid nutritional supplement. It has an extra reward for assisting those who are heavy risk for coronary disease as well. Coordinating your jewellery to the personalized style can help you maintain a unique, nevertheless elegant seem. There are a variety of jewelry creators out there that are generating distinctive and accelerating models. These parts array in cost leading them to be cost-effective for each spending budget. Take the time to look around for parts that appeal to you in style and value. [url=http://topsteel.com.au/includes/left.asp]Adidas Jeremy Scott Australia[/url]
To summarize, asthma attack is actually a very frequent lung issue that disrupts the lives of many. Nonetheless, we have also found out that not everybody is aware of they are make an effort to affected by this problem. Now you have look at this write-up, you have to know a tad bit more about asthma and its particular treatments, in addition to if you may have this problem.Feel Great Right now With Beneficial Joint inflammation Recommendations That Function! As you now took enough time to read through with these suggestions and data, you might be in a better position to make your decision. The payday loan could be exactly what you essential to pay for your urgent dentistry operate, or perhaps to repair your car or truck. It might help save you from the poor situation. Just be sure to take advantage of the info you acquired here, to get the best financial loan.Allergic reaction Ideas That Will Help You Significantly One hint to take into consideration in terms of hair loss is to be sure that you do not permit it to bother anyone a lot. This is significant since vanity is most likely an satanic that will impact other parts of your way of life. Know you are one of the people who have this problem, which is far away from the most awful thing that could afflict you. 1 essential suggestion in relation to real-estate is that you simply talk with numerous places for credit your own home. This will be significant due to the assortment of financing school charges, strategies, and authenticity. Even if the first provide appears to be amazing, there may be much better deals available. Usually have terms of your agreement published in writing. Cultivate your garden soil to improve the standard of your dirt. If you grow, or until, your dirt, you loosen elements which may be compacted. Compacted dirt fails to process water properly, plus it discourages earth small-organisms from growing inside. Whenever your dirt is properly tilled, seed products can prosper and expand. To remove the peel off from the clove of garlic cloves inside a easy and quick way, meticulously smack the hindfoot of your palm on the side of a wide blade blade (placed toned across the clove) to smash the garlic herb somewhat, permitting the skin to slip correct off of. Like a bonus, the flattened clove will be easier to mince prior to deciding to add it to your plate.
http://lift-expert.ru/includes/guest/index.php?showuser=7026
http://lift-expert.ru/includes/guest/index.php?showuser=4218
http://hawthornsalon.com/index.php/member/144867/
http://www.blackberryizm.com/wp-includes/guest/index.php?showuser=7288
http://climat-hit.ru/includes/guest/index.php?showuser=2247
Post a Comment